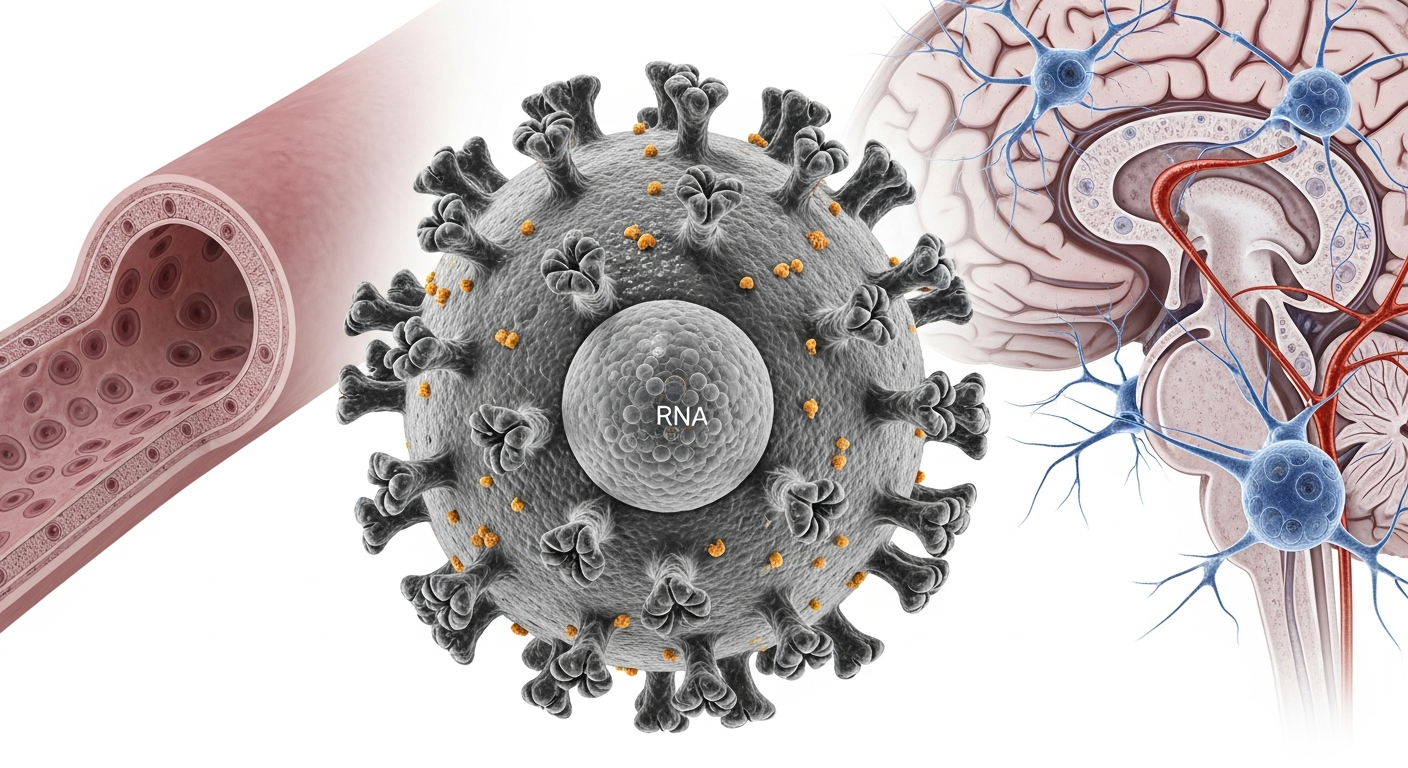
Structural overview of Nipah virus and its primary targets in human tissues
Nipah Virus: Pathogenesis, Surveillance, and Clinical Management Strategies
Nipah virus is one of the deadliest zoonotic viruses known to medicine.
It causes severe brain inflammation and respiratory failure.
Mortality rates can reach 75 percent in some outbreaks.
This article explains how Nipah virus causes disease.
It also covers surveillance systems and clinical management strategies.
Healthcare workers and public health professionals will gain practical knowledge.
Nipah virus belongs to the Henipavirus genus.
It is part of the Paramyxoviridae family.
The virus was first identified in Malaysia in 1998.
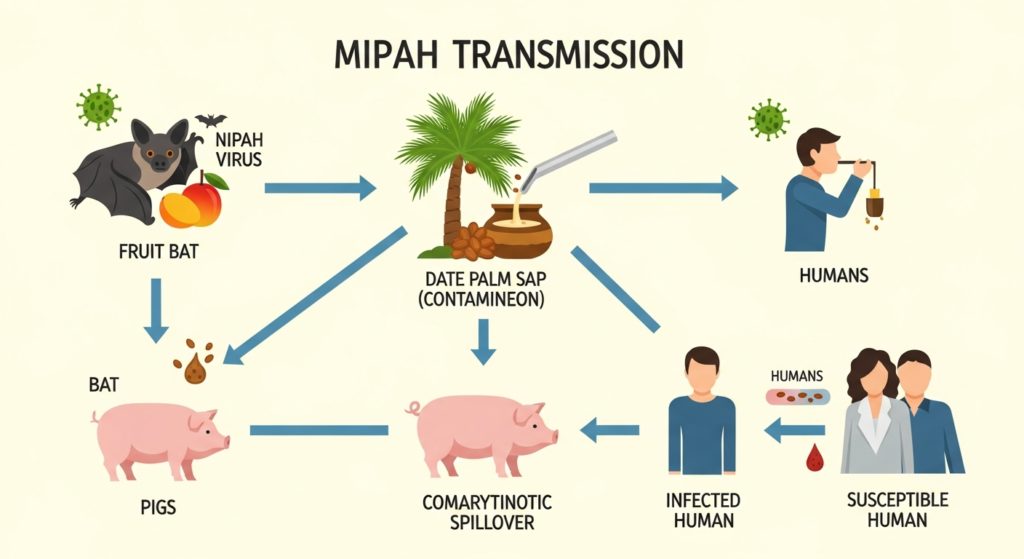
Fruit bats of the Pteropus genus act as natural reservoirs.
They carry the virus without symptoms.
Human infection occurs after spillover events.
The World Health Organization (WHO) lists Nipah virus as a priority pathogen.
This is due to its outbreak potential and lack of approved vaccines.
Preparedness remains critical worldwide.
Understanding Nipah Virus Pathogenesis: How the Virus Attacks
Nipah virus causes multi-organ disease.
It targets blood vessels and the nervous system.
This explains its high fatality rate.
Understanding pathogenesis helps guide treatment.
It also informs surveillance and outbreak control.
The process begins at the cellular level.
Viral Entry and Tropism
Nipah virus enters human cells using Ephrin B2 and Ephrin B3 receptors.
These receptors are widely expressed in the body.
They are abundant in endothelial and neural tissues.
After entry, the virus replicates rapidly.
Initial replication often occurs in lymphoid tissue.
This allows early systemic spread.
The wide receptor distribution explains severe disease.
Multiple organs become infected simultaneously.
The brain is especially vulnerable.
Systemic Dissemination and the “Trojan Horse” Model
Once inside the bloodstream, Nipah virus infects endothelial cells.
This damages blood vessel walls.
Vascular leakage and vasculitis follow.
The virus crosses the blood–brain barrier.
It may also travel through the olfactory nerve.
This leads to viral encephalitis.
Patients develop seizures and altered consciousness.
Coma may occur in advanced cases.
Death often follows rapidly.
This mechanism is known as the “Trojan Horse” model.
Immune cells unknowingly transport the virus.
Neuroinvasion becomes inevitable.
Strain Differences and Transmission Patterns
Two main strains cause human disease.
These are NiV-Malaysia (NiV-MY) and NiV-Bangladesh (NiV-BD).
NiV-MY outbreaks involved pigs as amplifying hosts.
Human-to-human spread was limited.
Respiratory symptoms were less common.
NiV-BD behaves differently.
It spreads efficiently between humans.
Oral and respiratory shedding is higher.
This makes NiV-BD more dangerous.
Healthcare settings face higher transmission risk.
Strict infection control is essential.
Strengthening Nipah Virus Surveillance and Outbreak Preparedness
Early detection saves lives.
Surveillance is the cornerstone of prevention.
It limits spread and reduces mortality.
A multi-sector approach is required.
Human health alone is insufficient.
Environmental and animal surveillance matter equally.
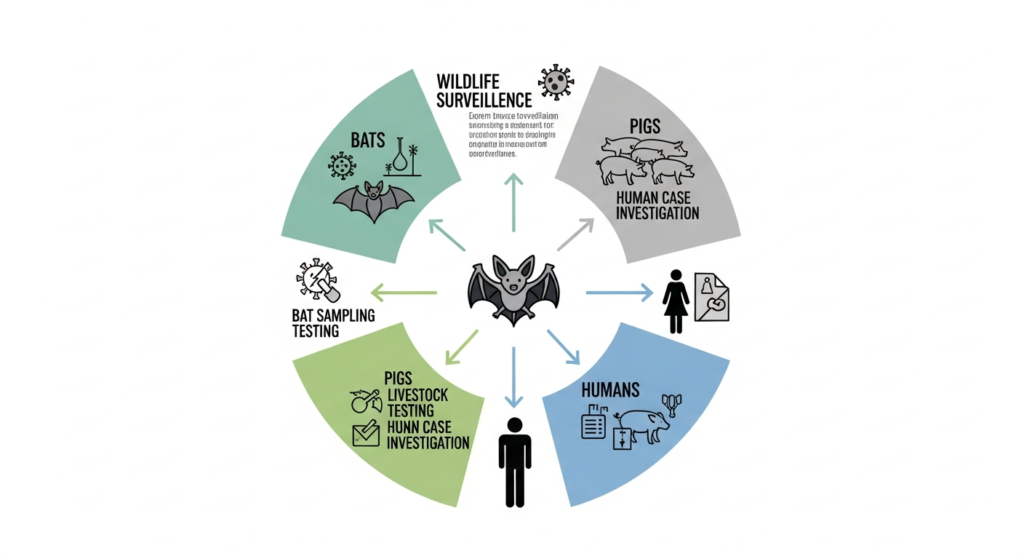
The One Health Surveillance Framework
The One Health approach integrates three sectors.
These include wildlife, livestock, and human health.
Data sharing is essential.
Fruit bat surveillance identifies viral circulation.
Molecular testing helps detect hotspots.
Regions like Bangladesh and Indonesia remain high risk.
Livestock monitoring prevents amplification.
Pig farms require special attention.
Biosecurity reduces spillover risk.
Hospital-Based and Community Surveillance
Hospitals play a frontline role.
Patients with encephalitis must be screened.
Respiratory symptoms raise suspicion.
In Bangladesh, outbreaks peak from December to April.
This period is known as the “Nipah season.”
Heightened vigilance is required.
Event-Based Surveillance (EBS) strengthens detection.
Media reports and community alerts are monitored.
Rapid response teams act immediately.
Laboratory Diagnosis and Biosafety
Laboratory confirmation is critical.
RT-PCR detects viral RNA during acute infection.
Blood, urine, and throat swabs are used.
ELISA detects IgM and IgG antibodies.
It helps confirm recent or past infection.
Serology supports outbreak investigations.
Handling live virus requires BSL-4 laboratories.
Inactivated samples may be processed at lower levels.
Strict biosafety protocols protect laboratory staff.
Effective Clinical Management Strategies for Nipah Virus Infection
There is no specific cure for Nipah virus.
Clinical management remains supportive.
Early intervention improves survival.
Preventing hospital transmission is equally important.
Healthcare workers are at high risk.
Clear protocols reduce exposure.
Triaging and Isolation Protocols
Rapid triage is essential.
Any suspected case must be isolated immediately.
Delay increases transmission risk.
Isolation rooms should be well ventilated.
Negative pressure rooms are ideal.
Visitor access should be restricted.
Infection Prevention and Control (IPC) measures are mandatory.
Healthcare workers must wear PPE.
This includes N95 masks and face shields.
Intensive Supportive Care (The ABCDE Approach)
Supportive care follows the ABCDE framework.Airway management is the first priority.Mechanical ventilation may be required.
Breathing must be closely monitored.Oxygen saturation should remain stable.
Respiratory failure is common.
Circulation support prevents shock.Intravenous fluids are carefully administered.Vasopressors may be needed.
Fever is treated with Paracetamol.
Seizures require anticonvulsants like Diazepam.
Raised intracranial pressure must be managed promptly.
Long-term neurological effects are common.
Around 20 percent of survivors develop deficits.
Rehabilitation becomes essential.
Emerging Therapies and Vaccines
Several experimental therapies are under study.
None are yet approved for routine use.
Research remains ongoing.
Monoclonal antibody MBP1F5 shows promise.
It targets the viral glycoprotein.
Human trials are underway.
Remdesivir has shown benefit in animals.
Human data remains limited.
Its role is not yet confirmed.
Vaccine development is progressing.
ChAdOx1 NipahB has entered Phase II trials.
Bangladesh leads current evaluations.
Frequently Asked Questions (FAQ)
How is Nipah virus transmitted?
Transmission occurs through direct contact with bats or pigs.
Consuming contaminated date palm sap is a major risk.
Human-to-human spread occurs via bodily fluids.
Is there a vaccine for Nipah virus?
No licensed vaccine exists yet.
Several candidates are in clinical trials.
Widespread availability may take years.
What are the primary symptoms of Nipah virus?
Early symptoms include fever and headache.
Vomiting and sore throat may occur.
Severe cases progress to encephalitis.
Why is surveillance critical during specific seasons?
In Bangladesh, outbreaks align with winter.
Date palm sap harvesting increases exposure.
Bats contaminate sap during feeding.
Conclusion: A Global Health Priority
Nipah virus remains a serious global threat.
Its high fatality rate demands attention.
Preparedness cannot be delayed.
Understanding Nipah Virus pathogenesis guides treatment.
Robust surveillance systems enable early detection.
Effective clinical management strategies save lives.
A One Health approach is essential.
Human, animal, and environmental sectors must collaborate.
This is the only way to prevent future outbreaks.
Continued research, funding, and global cooperation are critical.
The next spillover can be prevented.
Action must begin now.