Introduction
Alzheimer’s disease research has entered a critical phase. Several disease-modifying treatments now exist. However, most are given after brain damage has already begun.
Drugs such as lecanemab and donanemab focus on amyloid plaques. They are usually prescribed once memory problems appear. By then, neurons and synapses are already lost.
Now, a breakthrough from Northwestern University is changing this timeline. It introduces a new concept: NU-9: Stalling Alzheimer’s Through Early Astrogliosis Intervention.
This approach does not wait for symptoms. Instead, it targets silent brain changes that start decades earlier.
In this article, you will learn:
- What NU-9 is and how it works
- Why astrocytes are a hidden driver of Alzheimer’s
- How early intervention could prevent neurodegeneration
- What this discovery means for the future of dementia care
Understanding the Mechanism of NU-9: Stalling Alzheimer’s Through Early Astrogliosis Intervention
What Is NU-9?
NU-9 is a small-molecule compound, also known as AKV9 in scientific literature. Unlike antibody drugs, NU-9 is taken orally and can cross the blood–brain barrier effectively, making long-term preventive use more realistic.
Its primary function is simple but powerful: NU-9 blocks the buildup of toxic protein aggregates inside brain cells. These aggregates appear long before plaques form and silently disrupt cell function.
Current Alzheimer’s drugs require intravenous infusions and mainly target amyloid plaques already present. NU-9 works earlier and at a different biological level.
The Science of Reactive Astrogliosis
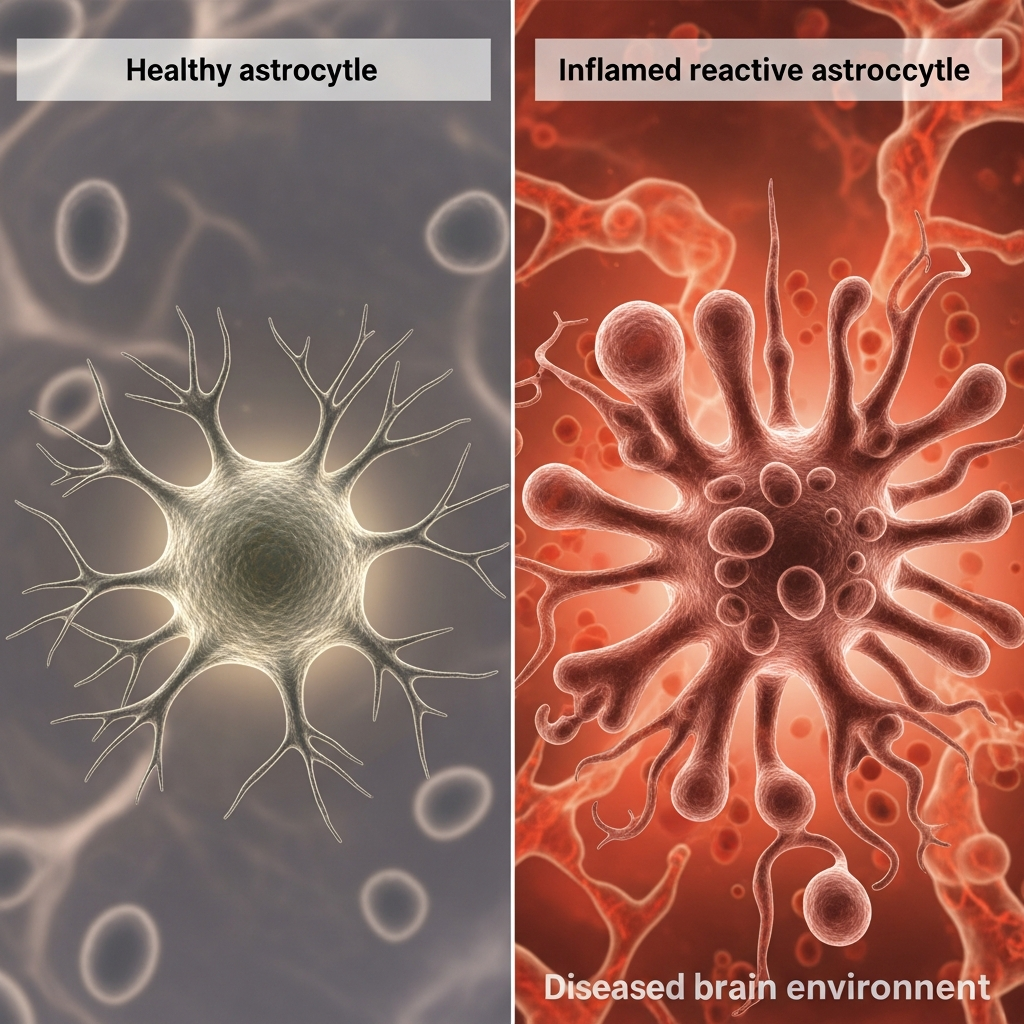
Astrocytes are star-shaped support cells in the brain. They protect neurons, maintain chemical balance, regulate inflammation, and support synaptic health.
In early Alzheimer’s disease, astrocytes become reactive and inflamed, a process called reactive astrogliosis. Instead of protecting neurons, reactive astrocytes:
- Release inflammatory molecules
- Damage synapses
- Disrupt neuronal communication
This occurs before memory loss. NU-9 works by keeping astrocytes calm, preventing harmful inflammation and preserving neuronal networks at a very early stage.
Targeting the “Hidden Culprit”: ACU193+ and Early Astrogliosis Intervention
Identifying the ACU193+ Subtype
Amyloid beta exists in multiple forms with different toxicity levels. Researchers identified a new subtype called ACU193+, which is highly toxic and appears very early in Alzheimer’s progression.
ACU193+ oligomers attach directly to astrocytes, triggering reactive astrogliosis. Unlike plaques, these oligomers are hard to detect but may cause most early brain damage. This shifts the disease model: Alzheimer’s may start with astrocyte dysfunction, not neurons alone.
How NU-9 Neutralises the Threat
NU-9 targets ACU193+ toxicity at multiple levels:
- Reduces toxic protein clusters inside neurons
- Lowers oligomer binding on astrocyte surfaces
- Restores natural cellular cleanup systems
This dual action stops inflammation before it spreads and prevents a cascade of damage, making early astrogliosis intervention possible.
Evidence from Northwestern: NU-9 Alzheimer’s Drug Success in Pre-Symptomatic Models
Stunning Results in Animal Trials
Northwestern researchers tested NU-9 in mouse models mimicking early Alzheimer’s pathology. Mice received oral NU-9 for 60 days. Results showed:
- Major reduction in brain-wide neuroinflammation
- Calmer astrocyte activity
- Improved cellular protein clearance
NU-9 also reduced abnormal TDP-43 protein, linked to cognitive decline in Alzheimer’s and other dementias.
Why the Pre-Symptomatic Window Matters
Most Alzheimer’s trials fail because they begin too late. By the time symptoms appear:
- Neurons are already dead
- Synapses are lost permanently
- Inflammation is widespread
NU-9 targets the disease before symptoms begin, interrupting the inflammatory chain reaction early and preserving brain structure for decades.
The Future of NU-9: Stalling Alzheimer’s Through Early Astrogliosis Intervention
The “Cholesterol Comparison” for Brain Health
Think about heart disease prevention: doctors prescribe statins before heart attacks happen. NU-9 may serve a similar role for the brain, used when biomarkers first appear.
Future care may include:
- Blood tests for toxic oligomers
- Early imaging markers
- Preventive NU-9 therapy
This shifts Alzheimer’s care from crisis management to prevention, potentially reducing global disease burden.
Transitioning from ALS to Alzheimer’s Clinical Trials
NU-9 has already cleared FDA approval for ALS trials, demonstrating acceptable safety for human use. For Alzheimer’s, next steps include:
- Testing in late-onset animal models
- Long-term safety evaluation
- Human pre-symptomatic trials
Conclusion
NU-9 represents a major shift in Alzheimer’s research. It focuses on early astrogliosis intervention rather than late-stage plaque removal. By calming reactive astrocytes, NU-9 protects neurons early and targets toxic oligomers before irreversible damage occurs.
While more research is needed, preventive neurodegenerative therapy is now scientifically plausible. Targeting astrocytes may become a cornerstone of future dementia care.
Frequently Asked Questions (FAQ)
What is reactive astrogliosis in Alzheimer’s?
Reactive astrogliosis is an early inflammatory response where astrocytes become overactive and damage synapses, occurring long before memory loss.
How is NU-9 different from Leqembi?
Leqembi targets amyloid plaques in early symptomatic patients. NU-9 targets toxic oligomers and astrocyte activation before symptoms appear.
When will NU-9 be available for humans?
NU-9 is already in human trials for ALS. Alzheimer’s trials require further animal testing first.
Can NU-9 reverse memory loss?
Current research focuses on prevention. NU-9 aims to stall disease before memory loss begins.
Analogy for Understanding
Imagine the brain as a high-tech factory. Astrocytes are the maintenance crew. Toxic protein clusters trigger a false alarm, and the crew panics, damaging machinery. This is reactive astrogliosis.
NU-9 acts like a calming specialist, silencing the false alarm early. The factory keeps running smoothly, and memory stays intact.